Explain How Atoms of Different Elements Are Different
Explore examples of elements and atoms. The different mix of energy differences for each atom produces different colours.
Difference Between Elements And Atoms Difference Between
Atoms combine in a ratio of small whole numbers to form compounds.
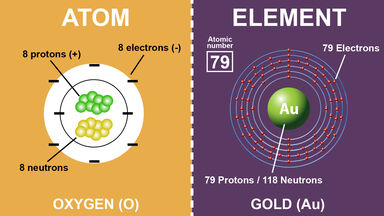
. However atoms and elements do have a few differences when you start breaking it down. Answer 38 5 9 mynameisnotmackenzie Each element is made up of one or more atoms. Learn other differences between atoms and elements by dissecting these two terms.
1 Explain how atoms of different elements differ from one another use Carbon vs Nitrogen 2 Explain how atoms of different isotopes of the same element differ from one another eg Carbon 12 vs Carbon 14. Isotopes are atoms that have the same number of protons in their nuclei but different numbers of neutrons. But the number of.
During a supernova the star releases very large amounts of energy as well as neutrons which allows elements heavier than iron such as uranium and gold to be produced. The nucleus in the center is surrounded by clouds of electrons. Some form specific types of bonds whereas others do not.
The atoms of different elements have different masses. Atoms are different from the total number of elements in a compound Advertisement khyroc08 is waiting for your help. An element is the simplest form of a.
This is the primary difference between an atom and element. An atom is the smallest particle of an element that retains the properties of that element. 2 Explain how atoms of different isotopes of the same element differ from one another eg Carbon 12 vs Carbon 14.
The main difference is elements are made of atoms. Add your answer and earn points. In order for most atoms to become stable they must bond with other atoms that are available.
Protons are the positively charged particles in the nucleus and they are what decide what the atom is. The only orbitals available to fill are larger in size so that an electron attempted to be placed in one of these sees an attraction to the nucleus and a corresponding matching repulsion by all the electrons in the lower-energy compact orbitals. Each metal gives a characteristic flame emission spectrum.
C12 C13 and C14. Different elements have different melting and boiling points and are in different states liquid solid or gas at room temperature. How they combine is based on.
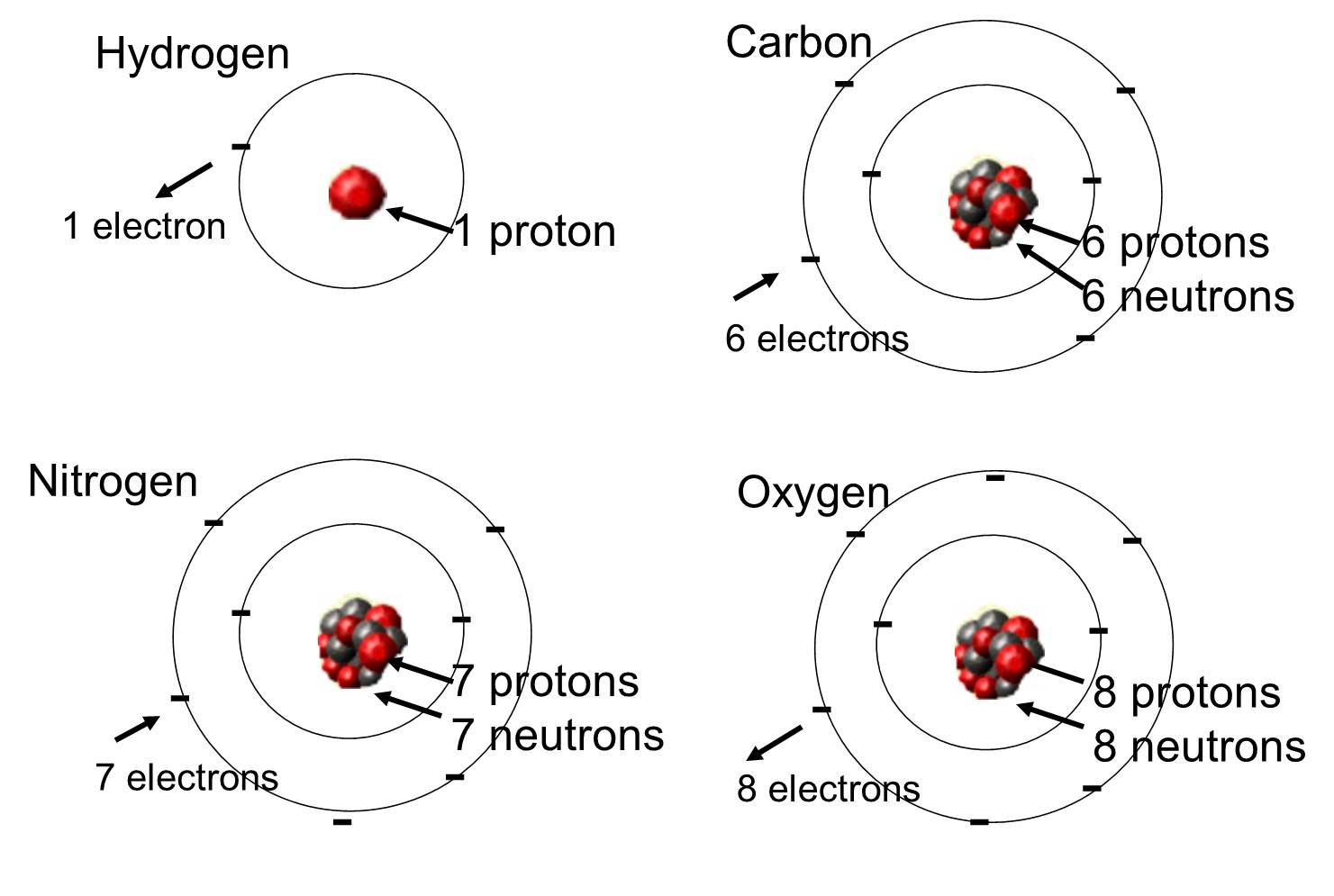
All atoms of an element have identical chemical properties and mass whereas atoms of different elements have different chemical properties and masses. When you heat an atom some of its electrons are excited to higher energy levels. These electrons are negatively-charged particles.
A compound is any molecule that is made up of two or more different elemental atoms. The elements formed in these stages range from oxygen through to iron. Therefore to be precise atoms are the smallest part or amounts of elements.
When an electron drops from one level to a lower energy level it emits a quantum of energy. An atom consists of two regions. A Atoms of different elements are different one another because of their different composition of protons electrons and neutrons.
Hydrogen has 1 proton helium has 2 protons lithium has 3 iron has 26 gold has 79 and so. In the supernova explosion all of these elements are expelled out into space. This problem has been solved.
The atomic number of an element refers to the number of protons in its nucleus and this number never varies - an atom with a different number of protons is a differentelement. They also combine in different ways. An element and an atom are different but related because a particular element is made up of many different types of atoms Cesar A.
1Explain how atoms of different elements differ from one another use Carbon vs Nitrogen. 6 rows Difference between Elements and Atoms. Atoms are the simplest unit of a matter.
12 Why atoms of the same element are the same. Determine whether the statement is true or false. By Staff Writer Last Updated March 28 2020 Elements primarily combine to form compounds through two main types of chemical bonding.
It has basic structural and fundamental units. For example the carbon within the human body is composed of mainly three isotopes. See the answer See the answer done loading.
Explain how atoms of different isotopes of the same element differ from one another. What makes one element different from another is the number of protons in the nucleus of its atoms. Many people might think atoms and elements are the same.
Ionic bonding and covalent bonding. The second much larger region of the atom is a cloud of electrons negatively charged particles that orbit around the nucleus. 11 How do different elements on it compare to one another.
In their center atoms have a closely packed nucleus. If false explain why it is false. The elements in the last family the column with He Ne Ar Kr Xe identify atoms with all the lower energy stabilizing orbitals completely filled.
13 When two atoms of the same element are bonded it is called a. The matter is anything and everything in our surrounding. Explain how atoms of different elements differ from one anotherb.
The first is the tiny atomic nucleus which is in the center of the atom and contains positively charged particles called protons and neutral uncharged particles called neutrons. Due to different number of these sub-atomic particles different elements have different physical and chemical properties and reactivity.
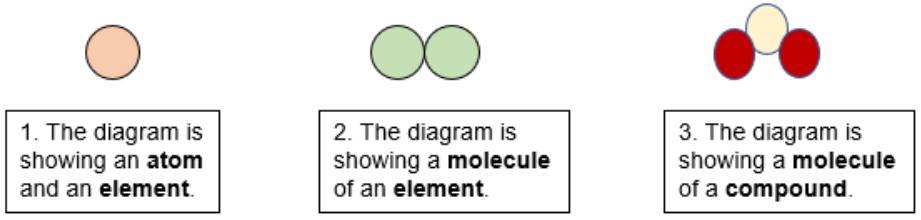
Understanding Atoms Elements And Compounds Lesson And Worksheets
Comments
Post a Comment